Image clarity is how well an imaging system can accurately reproduce the details and features of...
Decoupling Excitation & Detection: Radiometric Emitter Targets
In the world of fluorescence imaging, both excitation intensity and emission detection sensitivity are crucial for overall system performance. The QUEL Imaging Radiometric-Emitter Target (RET) allows users to de-couple these compounding factors. In brief, the RET is a calibrated solid-state emitter that emits known outputs at clinically-relevant wavelengths, enabling reproducible testing of fluorescence imaging systems. Among its many applications, this shelf-stable tool with controllable illumination helps investigate system collection efficiency of signals that mimic emission of clinically relevant fluorophores (e.g. ICG, Pafolacianine, SGM-101, or fluorophores at other custom wavelengths).
This tool is ideal for those who work with development and performance optimization of imaging systems and those conducting biomedical imaging studies, especially longitudinal imaging studies. We will break down the importance of both applications and how to implement them.
 |
 |
| Radiometric Emitter Target with custom controllable power supply. | Animation showing how multiple images of the RET can be combined to estimate an imaging system's collection efficiency. |
The RET enables analysis of capture efficiency of fluorescence imaging in multiple ways by mimicking fluorescence emission without requiring excitation, acting as an easy-to-use ground truth in:
- Stability and longitudinal imaging studies
- Characterizing fluorescence capture efficiency (i.e. collection efficiency)
Real-World Applications
Radiometric Emitter Targets offer versatility across system development, quality control, and research applications. Their stable and adjustable output makes them invaluable tools for both one-time system characterization and ongoing performance monitoring. Let's explore how the RET addresses key challenges in fluorescence imaging.
1. Stability and Longitudinal Imaging Studies
RETs serve as stable emission references during fluorescence imaging experiments to help maintain consistent detection sensitivity over time. As a stable reference, they can also be used to monitor degradation of fluorescent materials or consistent quantitative measurements between study locations. See the QUEL Imaging application note on monitoring system imaging performance and
photobleaching using the RET for more detailed guidance, but as an initial overview, let’s look at a few example scenarios:
Standardization
A researcher tracking fluorescence signals (i.e. from ABY-029) in an animal model over 8 weeks places an 800 nm RET at the same location in the field of view during each imaging session. The imaging system uses auto-gain to provide high contrast images. When the animal is positioned for imaging, the system's auto-gain adjusts until the RET signal reaches a predetermined pixel intensity value of 200. This constrains the imaging system’s auto-gain to provide consistent sensitivity across all time points, allowing for reliable quantitative comparison of fluorescence signals over the entire study duration.
Fluorescence Target Stability
A research lab uses an ICG-equivalent fluorescent phantom to validate their imaging system weekly, but the phantom went missing for a few weeks. They suspect the phantom was left under high-intensity illumination for an extended period. Luckily, when they first started using the ICG-equivalent phantom, they captured a fluorescence image of both their phantom and an 825 nm RET (set to power level 10) in the same field of view. Now they are able to capture the same image, and compare intensity values to determine whether a correction should be applied to account for small amounts of photobleaching, or if severe photobleaching occurred and the phantom needs to be replaced.
Multi-Center Clinical Trials
A multi-center clinical trial aims to quantify the dynamic flow of ICG. Each clinical site uses the same imaging system from a set working distance. Each site also receives an 825 nm RET with precise radiance characterization. The trial protocol requires imaging the RET at specific power levels before each patient procedure. Since the same imaging system and working distance was used, the RET images may be used to normalize the fluorescence measurements in each patient. This workflow provides a pathway to standardized quantitative comparison across different sites, ensuring that observed differences in fluorescence intensity represent actual biological differences.
2. Characterizing Fluorescence Capture Efficiency
RETs provide a stable reference to measure how much light the system collects. This is particularly valuable when evaluating optical collection efficiency. This de-couples the excitation uniformity from collection, providing data specific to how optical components channel light to the imaging sensor.
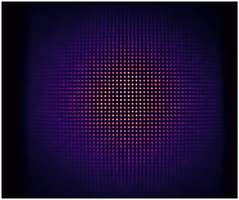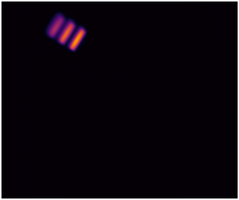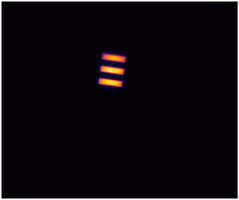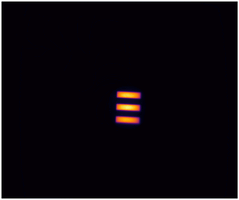
Example scenario: A manufacturer uses a 720 nm RET mounted on a stage to systematically map their entire field of view. By capturing images of the RET at multiple positions in grid pattern, like in the animation above, they generate the following relative illumination map.
 |
| Using the data collected in the animation above, the normalized intensity can be plotted across the field of view (left). These data points can then be interpolated and smoothed to visualize the relative collection efficiency (right). |
For detailed step-by-step instructions, follow Use Guide: Radiometric-Emitter Targets.
Best Practices for Development
The QUEL Imaging Radiometric Emitter Target is a robust tool for mimicking fluorescence emission. Given their precise, controllable, and stable radiance output, our RETs enable optimization of the fluorescence capture of an imaging system. Consider the following to help ensure your system meets clinical needs:
- What fluorophore emission wavelength do you need to mimic? Consider whether a standard RET (available options are: 720 nm, 800 nm, 825 nm) or if a custom wavelength is more suitable to match the specific fluorophore in your experiments.
- How will you account for external illumination effects on RET stability? Consider whether your RET will be imaged alongside fluorescent samples requiring excitation light, and how you will evaluate and mitigate potential illumination stability issues.
- What level of radiance precision do you require for your application? Consider whether you need the finer voltage control of the wired version (Appendix 1 in the Use Guide) or if the 20 discrete levels of the powered version are sufficient for your quantitative measurements.
- How will you ensure consistent positioning of the RET across multiple imaging sessions? Consider developing a jig or reference system if you're using the RET for longitudinal studies to minimize variation from spatial non-uniformity.
- What is your protocol for monitoring RET output over time? Consider how frequently you'll need to verify the RET's stability and whether you should schedule annual re-characterization to ensure ongoing measurement accuracy.
Test your system using the same settings (camera exposure time, camera gain, working distance, ambient lighting conditions, etc.) that will be used in clinical practice. If your system has different operating modes, characterize each one separately. By understanding these principles and following proper testing procedures, you'll be better equipped to develop and validate your fluorescence imaging system's capture efficiency.
Interested in characterizing your imaging system or developing a customized fluorescence reference target? Contact QUEL Imaging!